Electrophilic Substitution Reaction
What is Substitution Reaction?
In substitution reaction, one of the groups attached to the carbon atom of chain is replaced by another group. Below is a schematic representation of the substitution reaction.
Conditions for Electrophilic substitution reaction
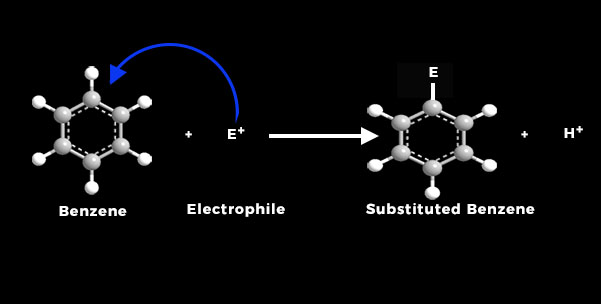
Electrophilic substitution reaction occurs on aromatic rings, because there is a delocalized π-electron cloud on aromatic compounds. Due to de-localization, each carbon atom in the aromatic ring has higher electron density as compared to the carbon atom on an aliphatic compound.
An electrophile is the positively charged species that is it is attracted towards electrons or an element that has higher electron density.
Higher Electron Density on Arene carbon atom
Aromatic compounds as discussed, have higher electron density than carbon atoms of aliphatic compounds. So aromatic compounds show electrophilic substitution reactions.
Mechanism of Electrophilic substitution Reaction
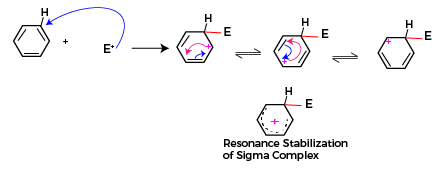
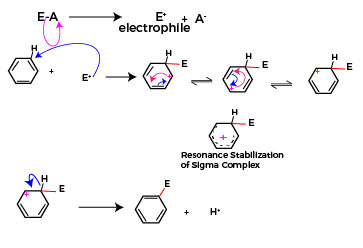
In this reaction, the first step is the formation of electrophile, the reagent undergoes heterolytic fission to form an electrophile.
The formed electrophile attacks the aromatic ring and correspondingly the intermediate formed is resonance stabilized. The resonance hybrid structure for the resonating forms of carbonium and in the benzene ring is shown below. This is the rate-determining step, and it is purely dependent on the stability of the carbonium ion
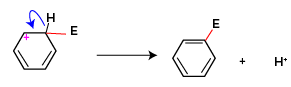
In the final step the carbon atom of the carbonium ion displaces the hydrogen and substituted benzene is formed as the final product. This displacement occurs because hydrogen is less electronegative than a carbon atom, carbon withdraws electrons and stabilizes itself and hydronium ion is displaced. This hydronium ion formed joins with the anion part of the reagent to form a byproduct during the electrophilic substitution reaction.
Common electrophilic substitution reaction
| S. No. | Reaction | Purpose |
|---|---|---|
| 1 | Chlorination Reaction | To introduce chlorine on aromatic ring |
| 2 | Bromination Reaction | To introduce bromine on aromatic ring. |
| 3 | Nitration Reaction | To introduce nitro group on aromatic ring. |
| 4 | Sulphonation Reaction | To introduce sulphonic acid group on aromatic ring |
| 5 | Friedel Craft Alkylation Reaction | To introduce Alkyl group on the aromatic ring. |
| 6 | Friedel craft acylation reaction | To introduce acyl group on the aromatic ring. |
All the above-mentioned reactions follow electrophilic substitution reaction. The general mechanism for these reactions is as below
You May also Like from Analytical chemistry:
| Ammonium Chloride | Preparation of 0.5 M Oxalic Acid | NCERT Class 11 Part 1 | Part 2 |
| Ammonium Nitrate | Preparation of 0.1 M Sodium bicarbonate | NCERT Class 12 Part 1 | Part 2 |
| Lead Acetate | Preparation of M/40 Mohr’s salt solution | CBSE Class 11 Syllabus |
| Aluminum Sulphate | CBSE Class 12 syllabus |
Electrophilic substitution reaction - Myetutors blog is a knowledge base of chemistry for IIT-JEE (mains), NEET-UG, CBSE exams.

Electrophilic substitution reaction helps in introducing a group onto the aromatic ring. The reaction is used extensively for preparing a variety of compounds
Course Provider: Organization
Course Provider Name: myetutors
Course Provider URL: https://www.myetutors.com
4.7
Electrophilic substitution reaction - Myetutors blog is a knowledge base of chemistry for IIT-JEE (mains), NEET-UG, CBSE exams.

Electrophilic substitution reaction helps in introducing a group onto the aromatic ring. The reaction is used extensively for preparing a variety of compounds
Course Provider: Organization
Course Provider Name: myetutors
Course Provider URL: https://www.myetutors.com
4.7