Acid catalyzed dehydration of alcohols
Cracking Organic Chemistry: Top 5 Tips to Master Acid Catalyzed Dehydration of Alcohols.
A blog post that provides valuable insights and study tips for students struggling with this complex concept in organic chemistry.
Introduction
Acid catalyzed dehydration of alcohols involves the elimination or removal of water molecules (H₂O) from alcohol molecules. When this reaction is applied to alcohols, dehydration yields alkenes. This process is typically aided by acidic catalysts or carried out in presence of mineral acid
Mineral Acids
In the acid-catalyzed dehydration of alcohol to alkene, a potent acid, such as sulfuric acid (H₂SO₄) or phosphoric acid (H₃PO₄), acts as the catalyst. The removal of water is achieved at reflux conditions.

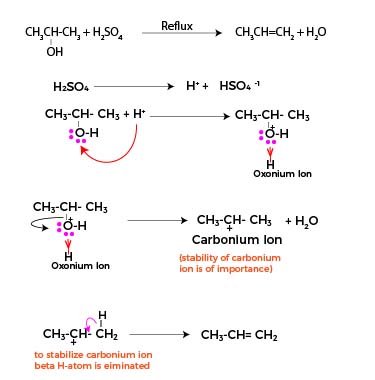
This reaction follows a set of steps:
Protonation of the Alcohol:
The acid protonates the alcohol’s hydroxyl group, resulting in an oxonium ion (R-OH₂⁺ ).

Formation of a Carbocation:
The protonated alcohol expels a water molecule, producing a carbocation as an intermediate.

Deprotonation to Generate Alkene:
The carbocation releases a proton, converting into the desired alkene, propene.

Advantages of Acid-Catalyzed Dehydration
- Enhanced Efficiency: Acid catalysts expedite the reaction by reducing the activation barrier.
- Precise Control: The use of strong acids enables better management of reaction conditions, leading to increased yields of the desired alkene.
- Reflux Condition: Reflux is commonly employed in dehydration reactions to maintain a consistent temperature, ensuring that the reaction mixture does not dry out. This condition brings about the following benefits:
- Stable Temperature: It sustains the reaction at a constant temperature, preventing overheating or underheating.
- Complete Reaction: Reflux guarantees that the reaction proceeds to completion by continually reacting the reactants until they are fully converted
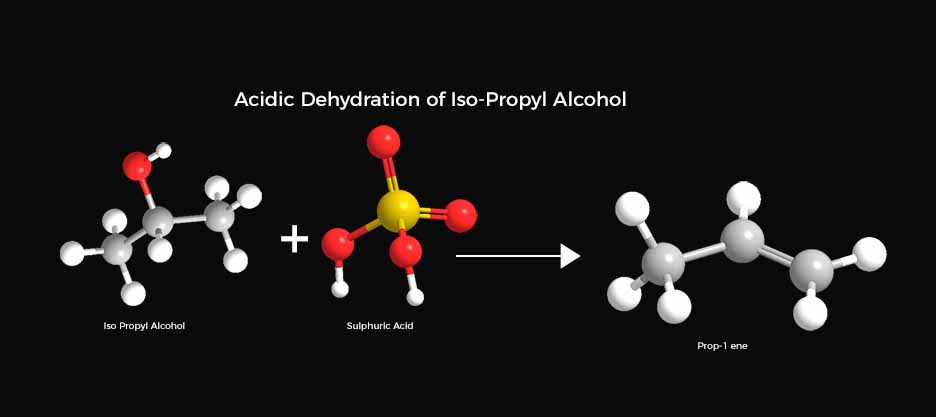
Illustration
Dehydration of Iso-Propyl Alcohol into Propene
Preparation:
A round-bottom flask is equipped with a reflux condenser. Iso-Propyl alcohol is mixed with a catalytic quantity of concentrated sulfuric acid.
Composition:
The concoction is subjected to a temperature that promotes dehydration, typically between 170-180°C. The reflux condenser effectively condenses any vapor and cycles it back into the flask, ensuring the desired volume of the reaction mixture.
Overall Reaction Mechanism
You May also Like from Analytical chemistry:
| Ammonium Chloride | Preparation of 0.5 M Oxalic Acid | NCERT Class 11 Part 1 | Part 2 |
| Ammonium Nitrate | Preparation of 0.1 M Sodium bicarbonate | NCERT Class 12 Part 1 | Part 2 |
| Lead Acetate | Preparation of M/40 Mohr’s salt solution | CBSE Class 11 Syllabus |
| Aluminum Sulphate | CBSE Class 12 syllabus |
