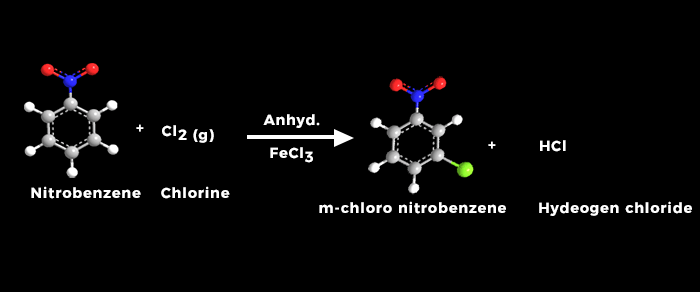
Chlorination of Nitrobenzene
The chlorination of nitrobenzene is an electrophilic substitution reaction, which involves the replacement of a hydrogen atom on the benzene ring with a chlorine atom. The reaction is typically carried out in the presence of a catalyst, such as ferric chloride (FeCl3), and a chlorinating agent, such as chlorine gas (Cl2).
Introduction to Electrophilic Substitution
Chlorination of Nitrobenzene is electrophilic di substitution, where an electrophile (an electron-deficient species) replaces a leaving group on arene molecule. In the case of aromatic compounds, the carbon atom is having higher electron density due to delocalization of π-electrons in aromatic ring.
The electrophilic substitution reaction involves three steps
- Formation of Electrophile
- Formation of Sigma complex.
- Stabilization of Sigma complex
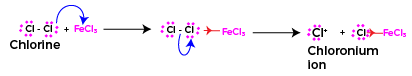
Formation of Electrophile
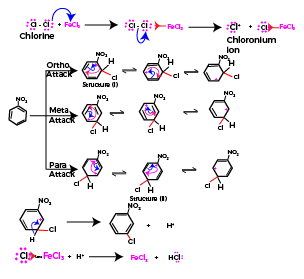
In the chlorination of nitrobenzene, the electrophile is the chloronium ion (Cl+), which is generated in situ from chlorine gas (Cl2) and a Lewis acid catalyst, such as ferric chloride (FeCl3) or Anhydrous AlCl3.
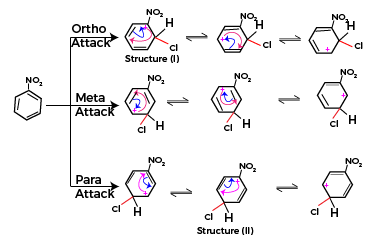
Formation of Sigma complex
The Chloronium ion formed as a reactive intermediate in the above step will join with the arene (here Nitrobenzene). Now when benzene is already having a substituent present on the ring the directive effect is applicable on the new incoming group (in this case chloronium ion as incoming group). The purpose of directive effect is to achieve the most stable end product where both already present group and incoming group are not inducing instability to the final structure.
Here nitro group is – I-effect (negative inductive effect) group and chlorine will join only on those positions where stability of nitro group is not decreased. The resonating forms for the substitution reaction are as below
In above resonating forms Structure (I) and structure (II) are exceptionally unstable structures. As positive charge is present on the carbon atom attached to an electron withdrawing group. Thus, the relative stability of meta-attack is higher than corresponding ortho and para-attacks.
Thus, incoming group will join at meta position on nitrobenzene.
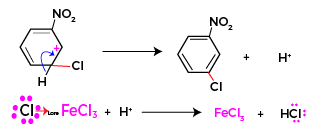
Reestablishment of aromatic character
The sigma complex formed loses a proton and aromaticity of arene is reestablished .
Chlorination of nitrobenzene one shot
The complete mechanism of chlorination of nitrobenzene in one shot is given as below
You May also Like from Analytical chemistry:
| Ammonium Chloride | Preparation of 0.5 M Oxalic Acid | NCERT Class 11 Part 1 | Part 2 |
| Ammonium Nitrate | Preparation of 0.1 M Sodium bicarbonate | NCERT Class 12 Part 1 | Part 2 |
| Lead Acetate | Preparation of M/40 Mohr’s salt solution | CBSE Class 11 Syllabus |
| Aluminum Sulphate | CBSE Class 12 syllabus |